CDMO 서비스
유연하고 협력적인 서비스
고품질의 GMP 제조 서비스를 제공함으로써 세포 치료제의 장벽을 돌파합니다.
위탁생산 소개
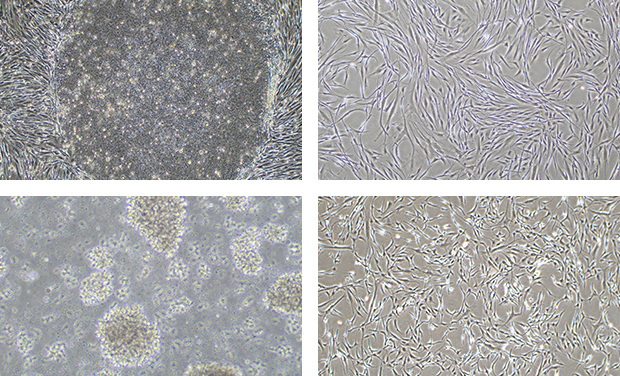
보유 기술 역량
- 10년 이상의 세포치료제 전문인력 다수 구성
- 다양한 세포 배양 및 분화, 증식 기술 보유
- 인체 유래 조직으로부터 세포 분리 및 동결 기술 보유

생산 Capacity
- 비임상시험용 의약품 생산
- 임상시험용 의약품 생산

GMP Facility 운영
- 독립형 제조시설 및 시험시설 보유
- 국내외 업허가 획득
-
 첨단바이오의약품
첨단바이오의약품
제조업 허가증 -
 일본 PMDA 해외제조시설
일본 PMDA 해외제조시설
인정 허가증 -
 인체세포등
인체세포등
관리업 허가증 -
 셀라토즈 세포처리시설
셀라토즈 세포처리시설
신고 승인 -
 의약품 제조업
의약품 제조업
허가증
시험 서비스 소개
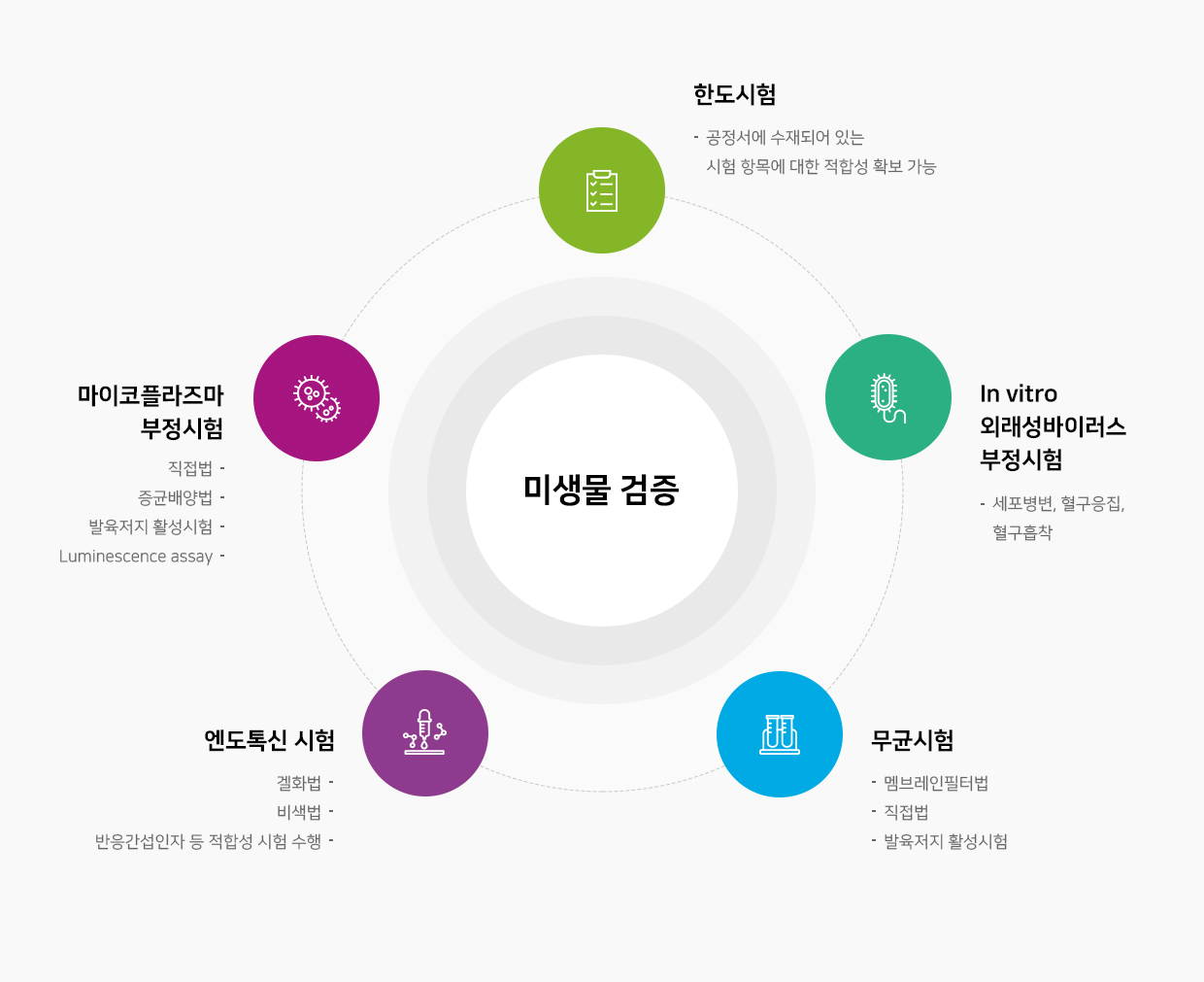
분석 시험 및 기타 검증 시험
-
 세포특성분석
세포특성분석- 세포 성장
- 세포 표현형
- 세포 분화
- 면역원성
-
 세포 안정성
세포 안정성 -
 기밀도 시험
기밀도 시험 -
 불용성이물 시험
불용성이물 시험
시설 소개

GMP 입구

GMP 입구

GMP 입구

일반시험실

일반시험실
일반시험실

일반시험실

일반시험실

일반시험실

일반시험실

바이러스실

바이러스실

세포보관소

세포보관소

패스박스

제조실

제조실

제조실

제조실

제조실

제조실

제조실

제조실

제조실

제조실

제조실

제조실

사무실 입구
PROCESS
-
01Pre-contract
-
02Process
Development -
03Technology
Transfer and
Pilot Runs -
04Process Assay
Development -
05Engineering
Runs -
06Aseptic
Process
Validation -
07Process
Validation -
08Production
CONTACT US
 gelim@cellatozrx.com
gelim@cellatozrx.com 070-5165-3974
070-5165-3974
Mon - Fri
8:00 - 17:00
8:00 - 17:00